Cardiovascular
- Research associate:
- Research assistant:
- Laboratory technicians:
Advanced ultrasound-based imaging and physiological monitoring for small-animal research are provided by the unit, offering comprehensive services for mouse and rat models. Originally focused on cardiovascular assessment, activities have been expanded to include oncology, neurology, metabolism, embryology, and general organ imaging. High-resolution anatomical and functional imaging, Doppler and 3D/4D modalities, photoacoustic imaging, and precise image-guided interventions are provided using the ultra-high- to low-frequency Vevo F2 LAZR-X ultrasound system.
Beyond ultrasound, ECG monitoring is performed in anesthetized, awake, or freely moving animals, and blood pressure is measured using tail-cuff and telemetric methods, alongside treadmill or pharmacological stress testing. Expertise covers preclinical studies, including imaging and functional assessment of embryos, neonates, and adults. Together, these technologies enable rapid, sensitive phenotypic screening and in-depth evaluation of cardiac and systemic physiology across diverse research fields.
Standard services Echocardiography (ECHO)
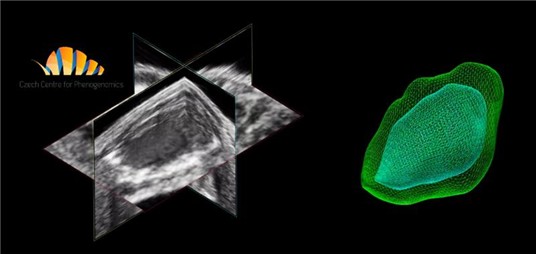
Echocardiography is used to monitor cardiac dimensions (chamber dimensions, thickness and valvular structures) and mechanics in vivo. For comprehensive evaluating cardiac structure and function we use standard two-dimensional B-mode, M-mode, EKV, PW Doppler and Tissue Doppler ultrasound or 3D/4D ultrasound. By using these techniques, we routinely perform:
- Ultrasound assessment of cardiac function in adult mice
- Left Ventricle Function
- Systolic and Diastolic Analysis
- Quantify cardiovascular function (ejection fraction, fractional shortening)
- 4D Analysis
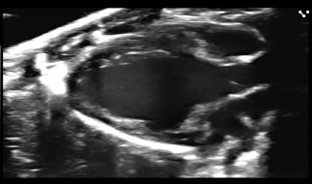
- Right Ventricle Function
- Wall thickness
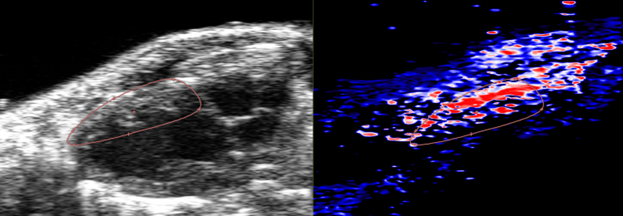
- Vessels Assessment
- Visualize vessel wall movement and vascular pathologies including atherosclerosis
- Assess blood flow through vessels and valves using Pulse Wave Doppler
- Analyze vessels with Vevo Vasc

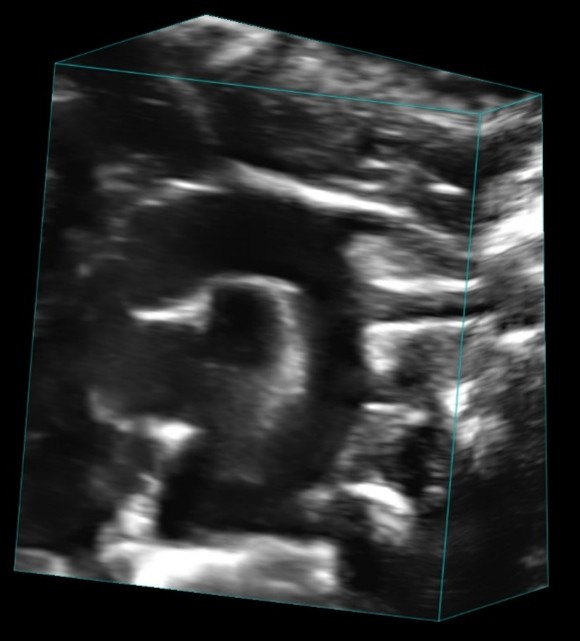
- Myocardial Wall Motion
- Quantify myocardial mechanics using Strain as an early biomarker for cardiovascular dysfunction
- Using Vevo Strain (ver.1 & ver.2)


The Vevo F2 ultrasound machine has three scan heads:
- UHF46x: 30 MHz with adjustable focal length providing an axial resolution of 50 μm – used in cardiac imaging in adult mice;
- UHF71x: 50 MHz with adjustable focal length providing an axial resolution of 30 μm – used in mouse vascular, abdominal, superficial embryonic imaging, epidermal and small mouse cardiac;
- UHF29x: 15 MHz – used in cardiac imaging of larger mice and rat, imaging of large tumors (< 23 mm) and nonlinear contrast imaging.
Echocardiography is typically performed in anesthetized mice (the system contains an anesthesia device that uses isoflurane); however, echocardiography is possible to do in conscious mice as well.
Standard services Electrocardiography (ECG)
Anesthetized ECG (aECG)
During echocardiography, the Vevo F2 ultrasound system routinely records murine cardiac electrical activity non-invasively through the animal’s paws using the Vevo Monitor system (VisualSonics). This setup provides a comprehensive ECG display based on up to 3-lead measurements, along with respiration rate and temperature monitoring. Animals remain under stable anesthesia throughout the procedure, ensuring consistent heart rate across all subjects.
ECG (awake)
The unit routinely records murine cardiac electrical activity non-invasively through the animal’s paws using the ECGenie system (Mouse Specifics, Inc.). The size and spacing of disposable footplate electrodes ensure optimal contact with the paws, enabling Einthoven lead II ECG acquisition in laboratory animals. For each subject, heart intervals and amplitudes are evaluated from continuous ECG recordings following a 5-minute acclimatization period. The system is equipped with ECG platforms and footplate electrodes of various sizes, allowing ECG monitoring of mice and larger rodents, including newborn pups.
ECG on Neonatal Mice
We use a non-invasive system (LifeSpoonTM; Mouse Specifics, Inc.) to characterize the postnatal maturation of the cardiac electrical conduction system of conscious neonatal mice. The system allows to monitor ECG abnormalities in newborn animals from day 1 of their life.
Long-Term ECG Telemetry (invasive)
Our unit employs the TSE telemetry system for continuous, long-term monitoring of cardiac electrical activity in rodents via implantable transmitters. This approach enables high-fidelity ECG acquisition in freely moving animals under physiological conditions, eliminating stress artifacts associated with restraint. The system supports multi-channel recordings, allowing precise evaluation of heart rate andintervals over extended periods. Telemetry platforms are available in various sizes, making them suitable for mice and larger rodents, and ideal for studies requiring uninterrupted cardiovascular monitoring during behavioral or metabolic experiments.
Analysis ECG
Our unit employs three dedicated software platforms: ECG Analyses (Mouse Specific), LabChart Pro (ADInstruments), and LabScribe ECG (iWorx). These tools enable precise acquisition and interpretation of cardiac electrical activity in small animal models. Typical parameters analyzed include heart rate, PR interval, QRS duration, QT/QTc intervals, and arrhythmia detection.
Standard services Blood Pressure
We provide accurate tail-cuff blood pressure measurement in mice using the CODA 8-channel High Throughput Non-Invasive Blood Pressure system (Kent Scientific). The CODA utilizes volume-pressure recording technology to detect changes in tail volume that correspond to systolic and diastolic blood pressures. Blood pressure measurements are made in conscious animals maintained in normal housing conditions with minimal handling and restraint of the animals. This reduces stress levels and physiological disturbances in blood pressure measurements over a longer period whereby data quality is improved. The unit is equipped with two CODA systems; thus, the measurement can be done on up to 16 mice or rats simultaneously. The method enables accurate blood pressure phenotyping in rodents for linkage or mutagenesis studies, as well as for drug testing experiments requiring high-throughput blood pressure measurements.
Do you have questions? Ask us
Custom services Stress Tests
Treadmill
Our treadmill system enables precise evaluation of cardiac function and exercise tolerance in mouse models under controlled stress conditions. Using the UGO Basile treadmill, we can simultaneously test up to six mice, applying adjustable speed and incline to mimic progressive workload. This approach is ideal for studying cardiac performance, endurance, and recovery in models of heart failure, ischemia, or hypertension, providing critical insights into cardiovascular adaptation and therapeutic efficacy.
Pharmacological
During echocardiography, mice receive dobutamine or another stimulantto induce cardiac stress and assess functional reserve. While the drug is active, cardiac performance is monitored using M-mode imaging (or alternative modes as required), providing precise measurements of ventricular function under increased workload. Simultaneously, ECG recordings are captured via the Vevo Monitor system (VisualSonics) to ensure comprehensive evaluation of heart rate and rhythm throughout the procedure.
Custom services Embryo Sonography (Fetal Sonography)
Gravidity check
Ultrasound confirms pregnancy in rodents from E6.5 onward, when external signs are not visible. We offer three levels:
- Standard – Basic check with 2D imaging.
- Counting – Check plus accurate embryo counting via 3D scan.
- Assessment – Full evaluation: vitality, counting, 3D imaging, Doppler.
Benefits: Early detection reduces animal use, prevents unnecessary euthanasia after miscarriage, and accelerates projects by enabling timely re-mating when pregnancy is not confirmed.
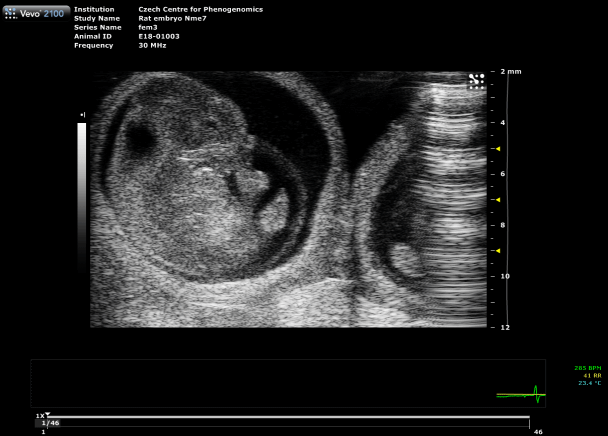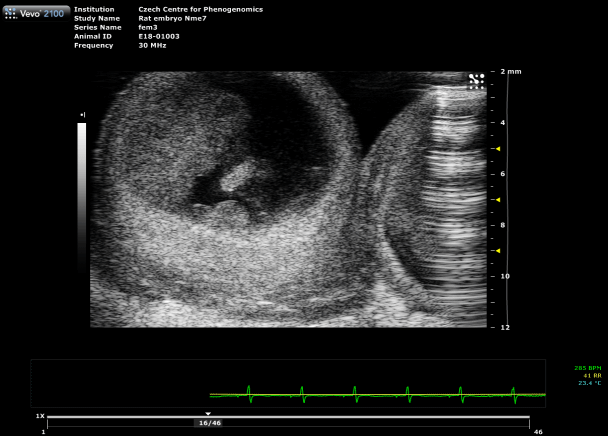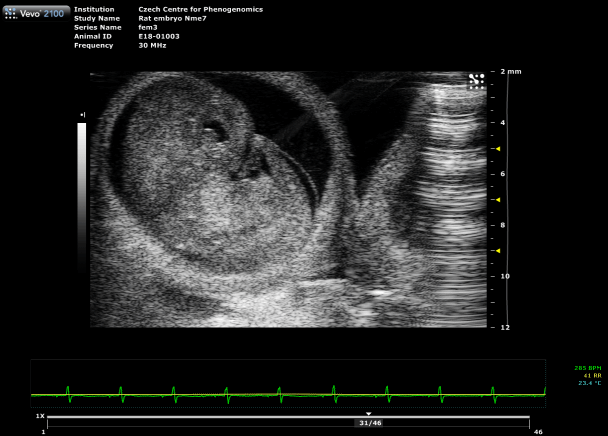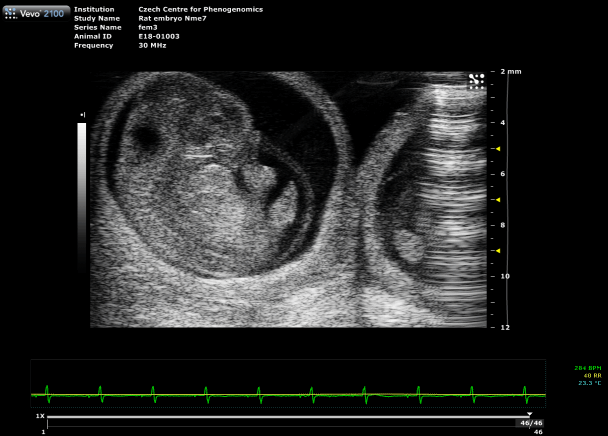
Placental Function Assessment
Using photoacoustic imaging, we measure oxygen saturation and hemoglobin concentration in the placenta to detect functional impairments. This method enables early identification of conditions such as preeclampsia, providing critical insights into fetal development and pregnancy health.
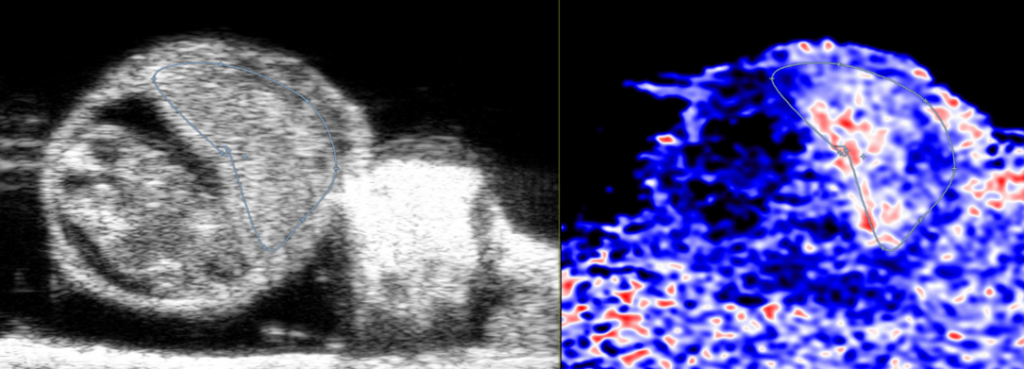
Custom services Oncology sonography (OncUS)
Using the Vevo F2 Lazr-X system, we monitor and quantify tumors in rodent models with high-resolution ultrasound and photoacoustic imaging. Our capabilities include:
- Morphological assessment: Tumor size and volume via 2D/3D imaging.
- Vascular analysis: Tumor vascularization with Power Doppler and blood flow measurement using PW Doppler.
- Advanced perfusion imaging: Nonlinear contrast with targeted or non-targeted microbubbles (Vevo MicroMarker) and quantification through Vevo CQ software.
- Photoacoustic imaging: Measurement of oxygen saturation and hemoglobin within tumors.
- Early detection: Photoacoustic contrast agents (wavelength 680–970 nm) for sensitive identification of small or early-stage tumors.
This comprehensive approach supports studies on tumor growth, angiogenesis, and therapy response.

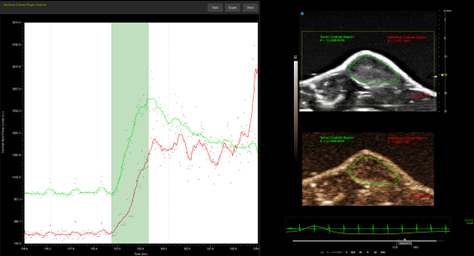
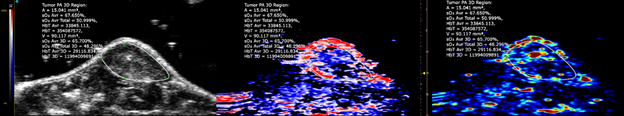

Custom services Cardiology
Myocardial Blood Saturation, Ischemia, Heart occlusion & Strain Analysis
Using the Vevo F2 Lazr-X system, we provide advanced assessment of myocardial oxygenation, ischemia, and cardiac mechanics in rodent models. Through photoacoustic imaging (PA mode) combined with EKV (Electrocardiogram-Gated Kilohertz Visualization), we measure oxygen saturation and hemoglobin concentration within cardiac tissue, enabling precise detection of ischemic regions. Additionally, we offer Vevo Strain analysis, a high-resolution speckle-tracking technique for quantifying myocardial deformation and contractility, providing deeper insights into cardiac function under physiological or stress conditions.
This integrated approach supports studies on cardiovascular disease progression and therapeutic interventions.
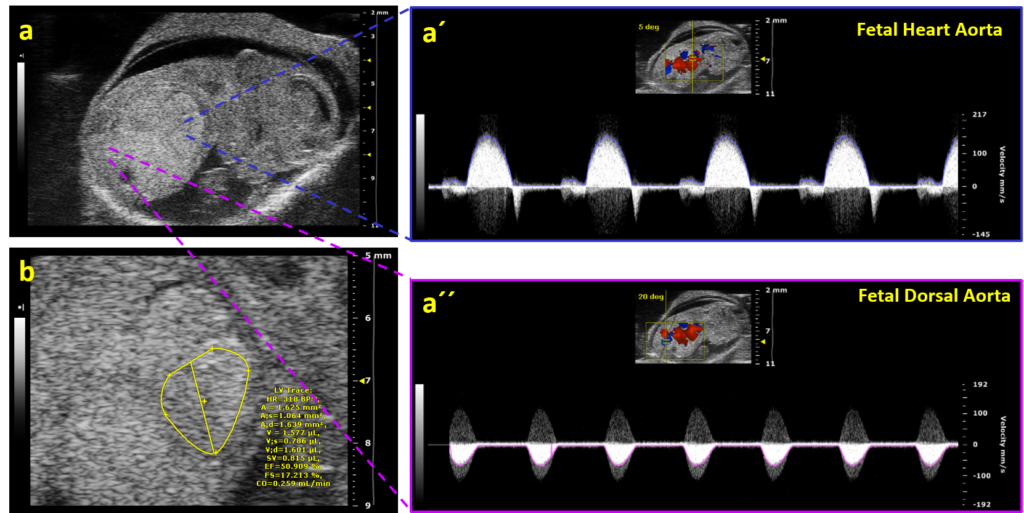
Fetal Echocardiography
For developmental studies, it is possible to monitor living mouse/rat embryos in uterus and follow the development of cardiac structures as well as changes in blood flow velocities in the heart and umbilical artery. An application of high-frequency probes with conventional 2D and pulse-wave Doppler imaging of the fetus can provide excellent information on the early development of cardiac structures.
Cardiomyocyte Isolation – Langendorff Method
We offer isolation of viable cardiomyocytes from rodent hearts using the Langendorff perfusion technique, a gold standard for ex vivo cardiac cell preparation. This method involves retrograde perfusion of the heart through the aorta with enzymatic digestion, ensuring high-quality single-cell suspensions of ventricular myocytes.
- Calcium imaging for intracellular Ca²⁺ dynamics and excitation-contraction coupling.
- Electrophysiological studies (patch-clamp, action potential analysis).
- Drug testing on isolated cardiomyocytes for pharmacological research.
- Molecular assays (gene expression, protein analysis) on purified cardiac cells.
This approach provides a controlled environment for studying cardiac physiology at the cellular level, complementing in vivo imaging and functional assessments.
Cardiomyocytes Contractility & Electrophysiology
We use the Agilent RTCA CardioECR system to monitor contractile function and electrical activity of neonatal cardiomyocytes in real time. This platform combines impedance-based measurements for contraction analysis with extracellular field potential recording, providing simultaneous insight into mechanical and electrophysiological properties of cardiac cells.
The system enables high-throughput, label-free monitoring under physiological conditions, making it ideal for preclinical research and compound screening.
Custom services Image-guided injection (IGI)
Using the Vevo F2 Lazr-X ultrasound system combined with a micromanipulator, infusion micro-pump, and nano-pump, we perform precise, minimally invasive delivery of cells, viral vectors, or drugs into specific organs under real-time imaging guidance. This approach ensures accurate targeting and reduces tissue damage compared to conventional methods. Example injection sites: Kidney, Myocardi, Pancreas, Tumor, Neonatal Brain, Prenatal (e.g. cerebral ventricles), intraocular and other custom locations upon request. This technique supports advanced experimental designs, including gene therapy, cell transplantation, and localized drug delivery.
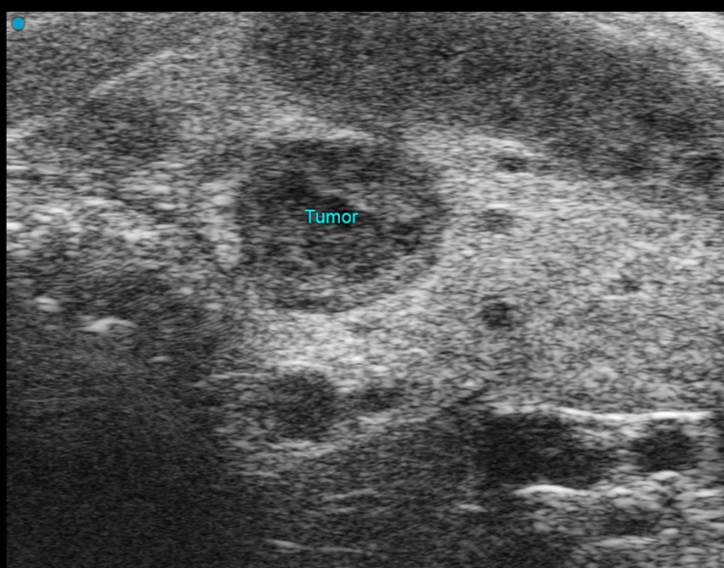
Custom services Neurology Sonography
Our Vevo F2 Lazr-X system, equipped with a stereotactic frame, enables high-resolution imaging of the rodent brain using multiple modalities:
- B-mode for structural visualization.
- Doppler imaging for monitoring cerebral blood flow and vascular changes.
- Nonlinear contrast imaging with microbubbles for perfusion studies.
- Photoacoustic imaging (PA) for measuring oxygen saturation and hemoglobin concentration in brain tissue.
These capabilities allow detection of vascular alterations, blood flow dynamics, and oxygenation changes, as well as monitoring tumor growth and perfusion patterns. For early tumor detection, photoacoustic contrast agents (wavelength 680–970 nm) can be applied. This integrated approach supports research in stroke, ischemia, neuro-oncology, and other neurological disorders.

Custom services Ophthalmology
Using high-resolution ultrasound, we monitor eye development from embryonic stages through adulthood in rodent models. Our services include:
- Volumetric measurements of ocular structures for growth and morphology studies.
- Vascular assessment using Doppler imaging to evaluate ocular blood supply, including the central retinal artery.
- Blood flow analysis with PW Doppler for functional insights.
- Intraocular image-guided injections (IGI) for targeted delivery of compounds or contrast agents.
This approach supports research in developmental biology, ophthalmic diseases, and therapeutic interventions.
Custom services General Sonography
Beyond specialized applications, we offer ultrasound-based assessment of multiple organs in rodent models, with options for structural and functional analysis:
- Liver – Monitoring of steatosis and fibrosis through structural imaging or lipid quantification using photoacoustic mode.
- Spleen – Evaluation of size, vascularization, and blood flow with Doppler techniques.
- Kidneys – Morphology and perfusion analysis, including renal blood flow measurements.
- Reproductive organs – Imaging of uterus, testes, and mammary glands for developmental or pathological studies.
Custom protocols can be designed for other organs or specific research needs, combining 2D/3D imaging, Doppler, and photoacoustic techniques for comprehensive evaluation.
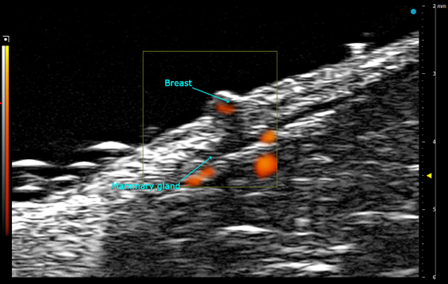
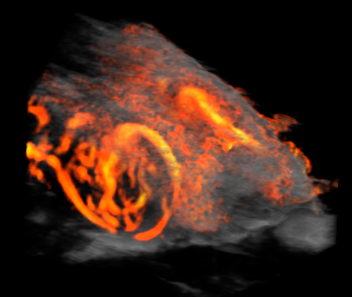

Custom services Ultrasound engineering
Our system features Vevo Advanced Data Acquisition (VADA), enabling custom-programmed acquisitions. The graphical user interface allows configuration of:
- Transmit Delays: plane wave, focused, custom, beam steering ±30°
- Waveforms: sinusoidal, chirp, pulse inversion, custom
- Pulse Sequences: walking aperture, plane wave (single or multi-angle), voltages, laser, 3D motor, triggers, and more
This is an excellent solution for developing and exploring new imaging methods.
Do you have questions? Ask us
Cardiovascular unit was upgraded and expanded with the support from OP RDE project CZ.02.1.01/0.0/0.0/18_046/0015861 CCP Infrastructure Upgrade II in the years 2020 – 2022 and currently it is being upgraded from the OP JAC project CZ.02.01.01/00/23_015/0008189 Upgrade of the large research infrastructure CCP III.